Carboxylic Acids old
Carboxylic acids can occur naturally :
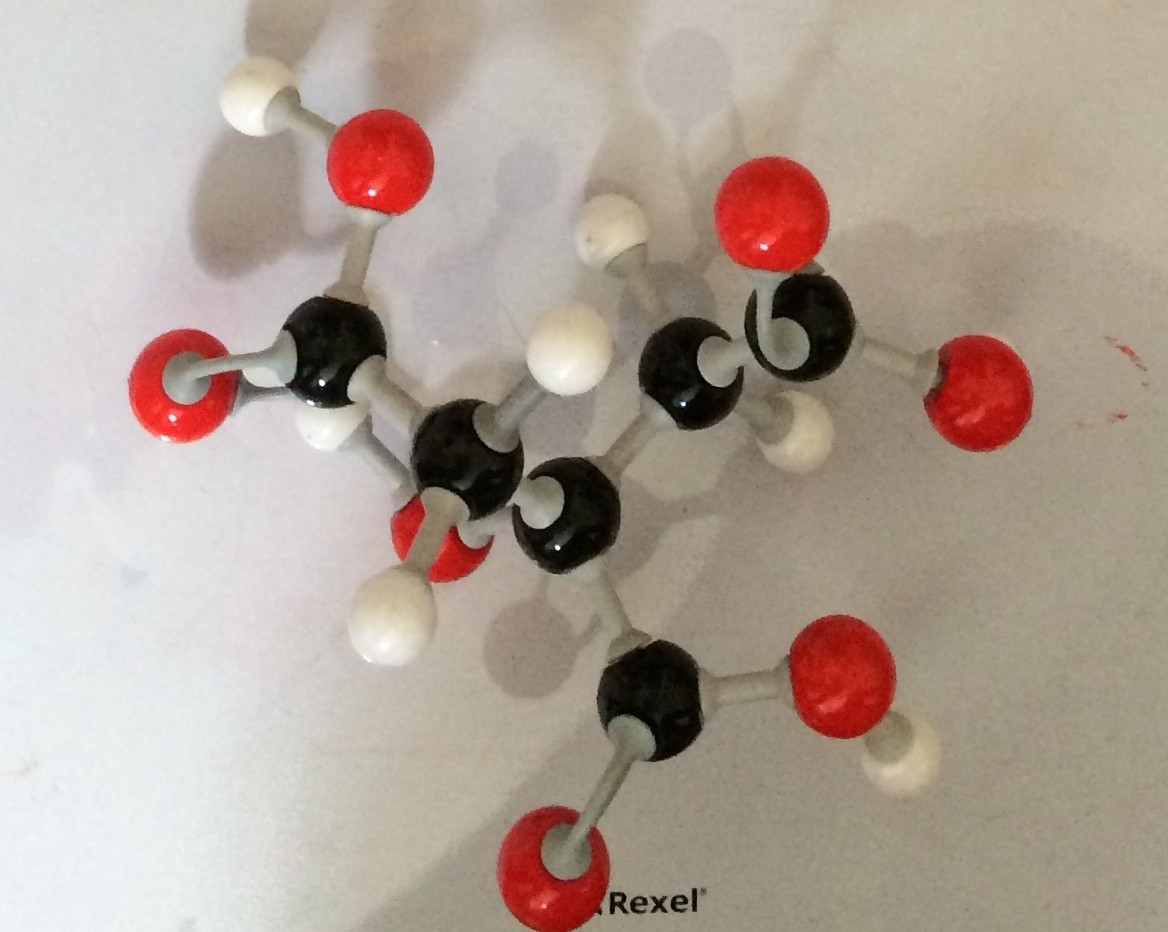
When alcohols are oxidised they can produce a type of acid known as a carboxylic acid.
one example of a carboxylic acid is ethanoic acid . This is also known as Acetic acid and is the acid found in vinegar.
Other examples of carboxylic acids are methanoic acid ( also known as formic acid) which is found in the sting of Ants and Bees.
If a bottle of wine is left open to the air for too long, the alcohol ( ethanol) in it gets oxidised ( to ethanoic acid) and can become acidic and tastes unpleasant.
Making carboxylic acids in the laboratory
We can make carboxylic acids in the laboratory by mixing an alcohol with an oxidising agent and heating it up. The first four minutes of this video show the apparatus used. Notice that the condenser is mounted vertically on top of the flask. Why do you think it is mounted vertically?
answer
Notice the colour changes which take place as the reaction proceeds.
Acids we eat
Making pineapple from vomit
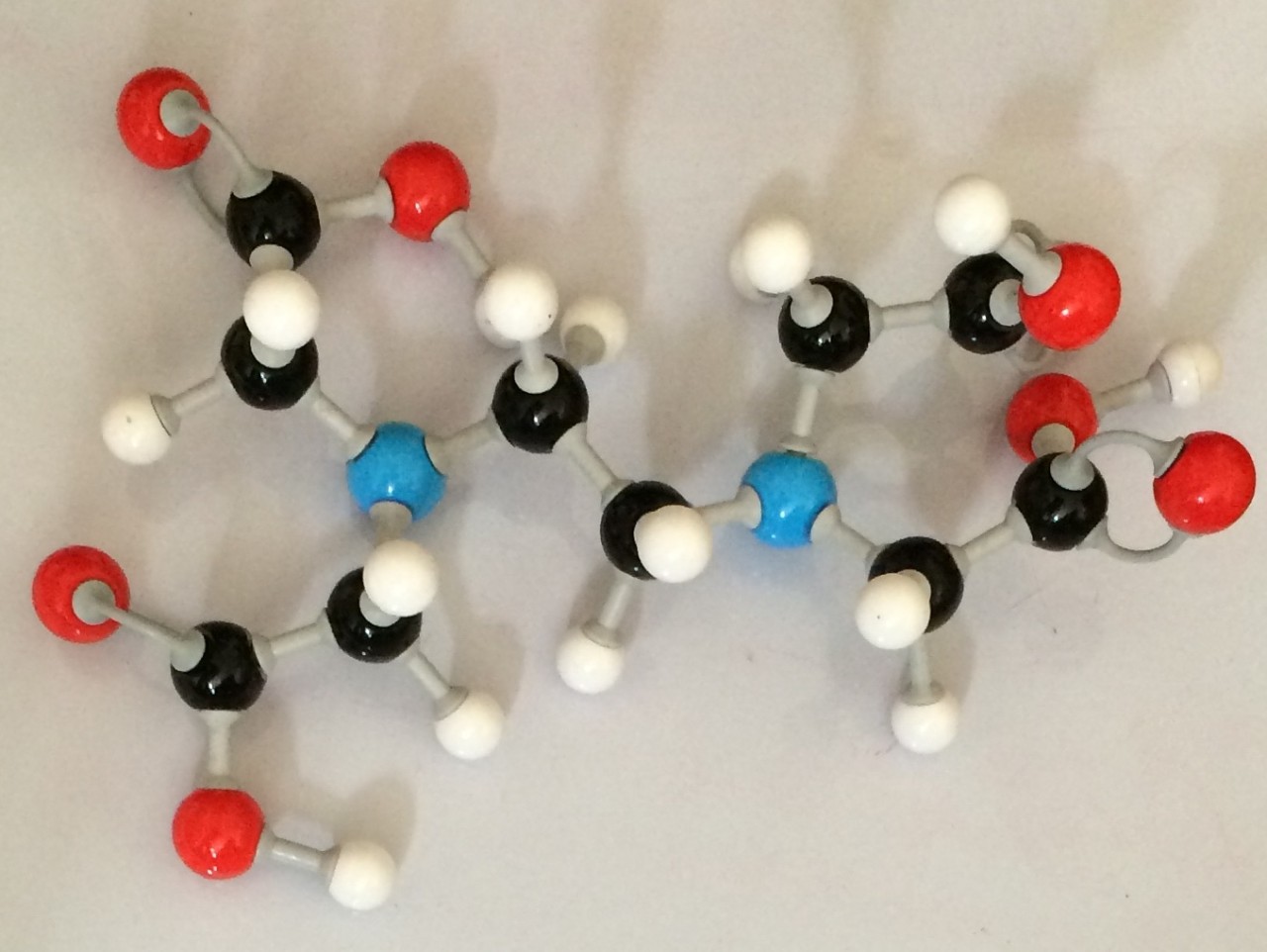
In this video you will see how a foul smelling carboxylic acid can be changed into the sweet smell of pineapple. The reaction involves a carboxylic acid combined with an alcohol to form an ester.
1. What is the name of the carboxylic acid that smells like vomit? :ethyl butanoate.
2. What is the name of the alcohol used? :ethyl
3. What other ingredient is needed to help the esterification reaction? :sulphuric acid
4. What apparatus is used to get the carboxylic acid to combine with the alcohol? condenser
5. What is the name of the product formed? an ester
6. What does this product smell like? pineapple
7. How is the product separated from the reactants? distillation
8. What is reflux? the process of boiling a liquid so that any vapour is liquefied and returned to the stock
9. What is distillation? distillation is separation of components of substances.
Name that Ester!
This animation from Richard Thornley explains how to name esters produced by the process above.
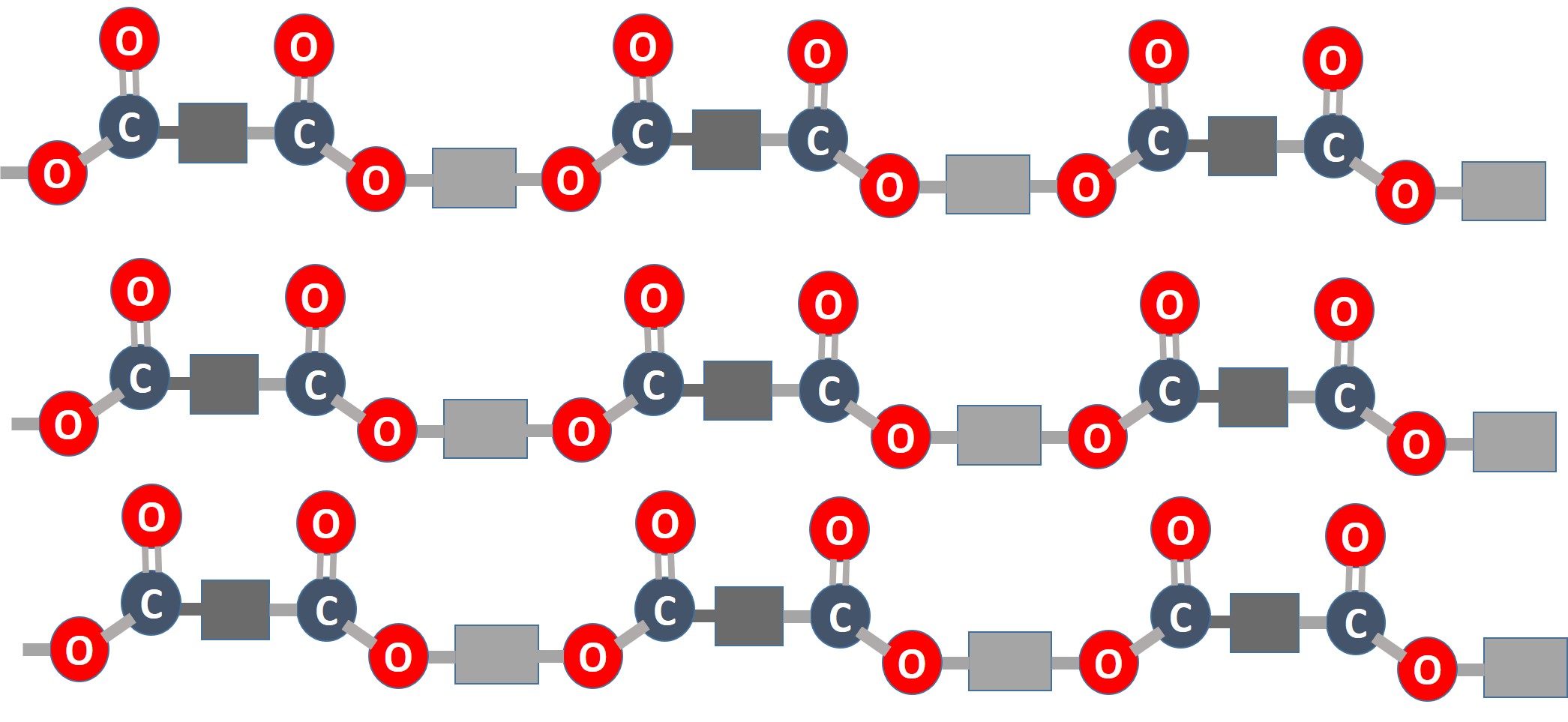
why are polyesters useful?
Polyester is often used in outerwear because of its high tenacity (strong and stretchy) and durability. It is a strong fibre and consequently can withstand strong and repetitive movements. Its hydrophobic (which means water hating) property makes it ideal for garments and jackets that are to be used in wet or damp environments. Coating the fabric with a water-resistant finish intensifies this effect. sails for boats are made like this. In the climbing environment a lot of the ropes used are made of polyester. To do this they need to be very resistant to all the stress that goes through them. After all they have to survive the stress of a man falling on them and being rubbed agenst the rock walls.
When you subscribe to the blog, we will send you an e-mail when there are new updates on the site so you wouldn't miss them.