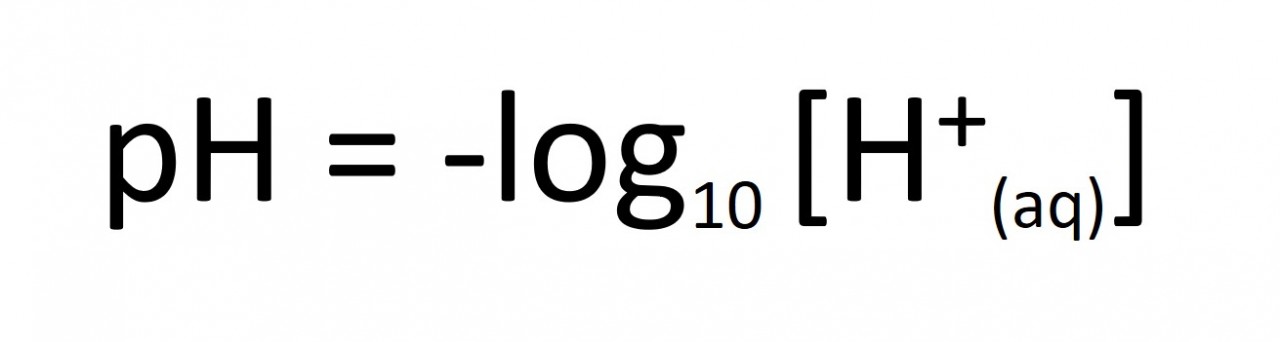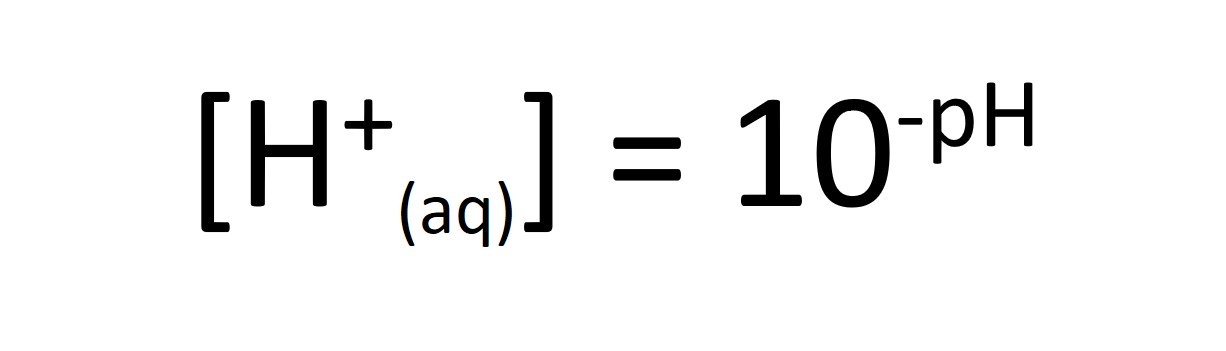Acids, Bases, pH, Indicators and Buffers
This topic is mostly about equilibria so it is important for students to understand that concept first. Before tackling this topic make sure you :
- understand dynamic equilibria
- can write and manipulate equilibrium expressions for a range of reactions.
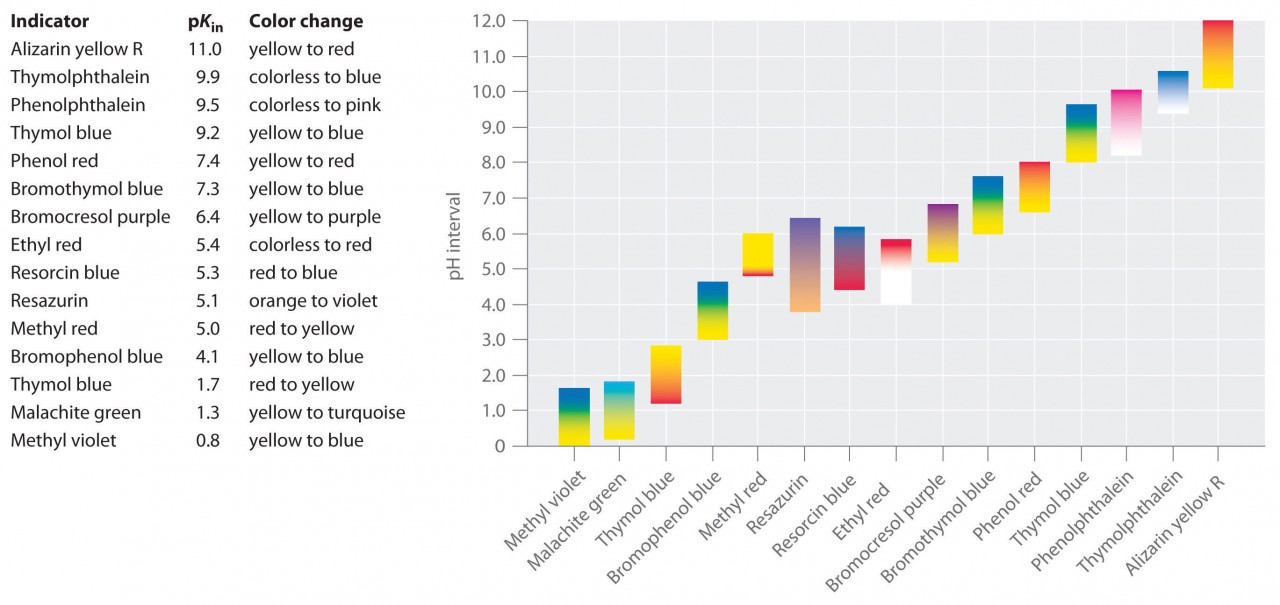
- can estimate the pH value of a solution using universal indicator
- know the pH values associated with : strong acids, weak acids, neutral solutions, weak alkalis and strong alkalis.
Strong acids vs weak acids
When an acid molecule ionises completely when in solution in water, it is regarded as a strong acid. It is said to dissociate completely.
If we consider this ionisation to be an equilibrium - it would lie completely to the right.
The equilibrium constant (Kc) would be a very large value and not really useful.
When calculating pH values for strong acids we assume that the concentration of H+ ions is equal to to the concentration of the acid.
Explaining buffers
Matt from chemistrystudent.com does a brilliant job of explaining buffer solutions here...
- 0:00 - 2:40 : Recap of essential concepts
- 2:40 - 11:00 : How buffers work
- 11.00 - 15:36 : Using buffers